Summary
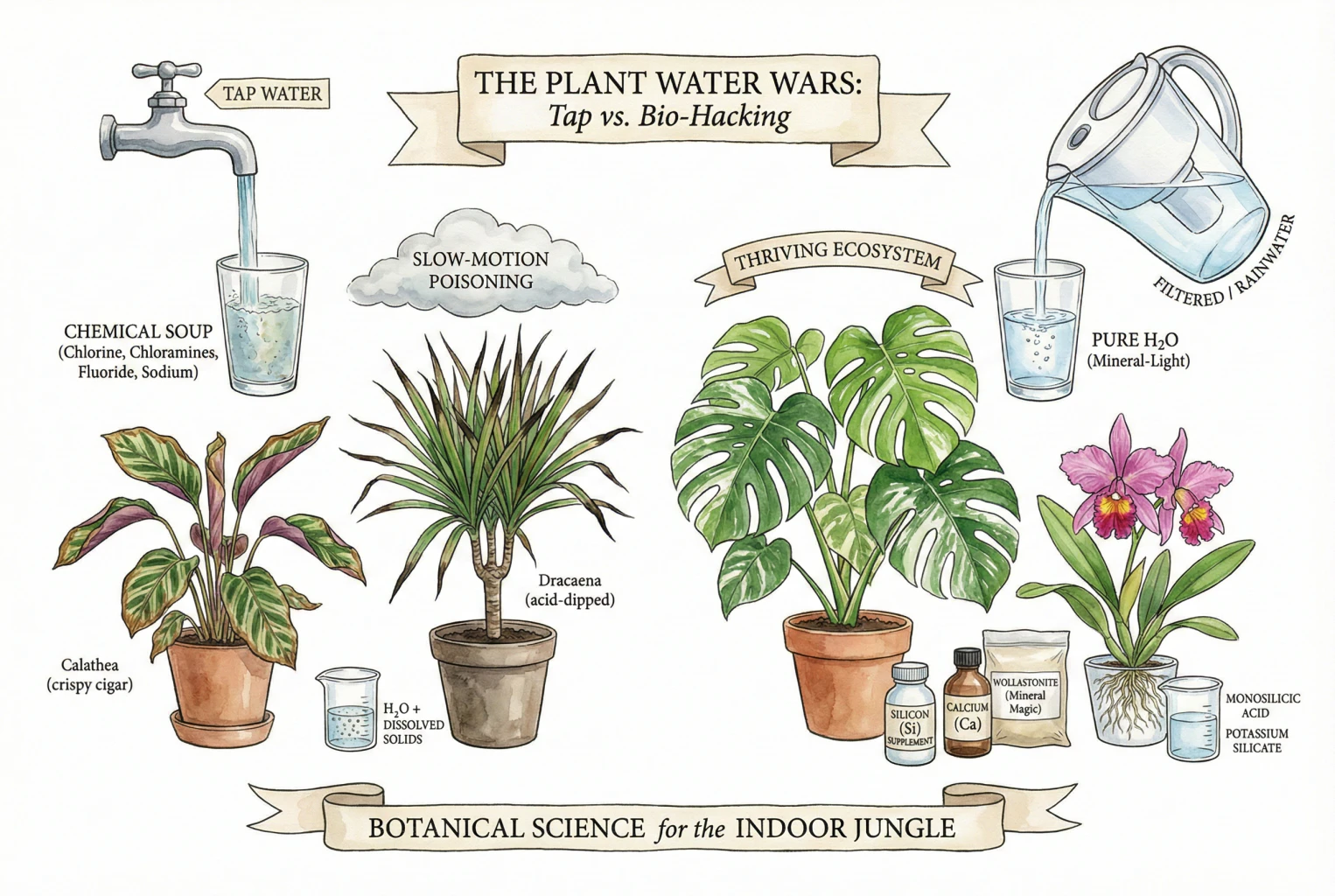
- Many common houseplant issues, such as brown leaf tips and stunted growth, are actually caused by invisible toxins in municipal tap water—specifically Fluoride, Chloramines, and Sodium—rather than simple underwatering or dry air.
- Recent scientific research highlights Silicon (Si) as a “missing link” nutrient that acts as biological armor, strengthening root barriers to block toxin uptake and boosting the plant’s antioxidant defense systems.
- Instead of relying solely on expensive filtration, using Wollastonite (Calcium Silicate) as a soil amendment offers a sustainable, low-maintenance solution to neutralize acidity, supply protective silicon, and ensure plant health.
Key Takeaways
- The “Invisible” Toxins:
- Fluoride: Accumulates in leaf tips, causing irreversible necrosis (browning), especially in sensitive plants like Dracaena, Calathea, and Spider Plants.
- Chloramines: Unlike chlorine, chloramines do not evaporate when water sits out; they actively kill beneficial soil microbiome.
- Sodium: Water softeners replace calcium with sodium, creating “physiological drought” where roots cannot absorb water even in wet soil.
- The Silicon Solution:
- Silicon is not just a structural element; it reinforces the Casparian strip in roots, acting as a physical bouncer to stop toxins from entering the plant.
- Monosilicic Acid (MSA) is the fastest-acting form for immediate rescue, while Potassium Silicate is a cheaper alternative that requires careful pH balancing.
- Wollastonite (The “Lazy” Expert Hack):
- Wollastonite is a naturally occurring mineral that breaks down to release both Calcium and Silicon.
- It acts as a slow-release buffer, preventing soil acidification (which makes fluoride more toxic) and continuously armoring the plant.
- Water Quality Hierarchy:
- S-Tier: Rainwater & Aquarium Water (Fresh, mineral-balanced).
- A-Tier: Reverse Osmosis (RO) + CalMag Supplement (Clean slate, remineralized).
- B-Tier: Tap Water + Dechlorinator + Silicon/Wollastonite (The practical standard).
- F-Tier: Softened Water & Untreated Greywater (High sodium/alkalinity risk).
Introduction: The Great Hydration Lie
You aren’t a bad plant parent; you’re just using the wrong water. Tap water is packed with chemicals that are safe for people but toxic to plants, and no amount of humidity will fix that.
We are going to ignore the marketing fluff and use recent scientific data to fix your water chemistry. By using specific minerals like Wollastonite and understanding Silicon uptake, we can stop the slow-motion poisoning of your ecosystem.
Part1: The Invisible Enemy — The Chemistry of Municipal Water

To defeat the enemy, you must know the enemy. And in the case of indoor gardening, the enemy is often disguised as a life-giving resource.
Municipal water treatment is a marvel of modern engineering, designed to deliver pathogen-free, palatable water to millions of people. But the very additives that make water safe for mammals prevent it from being the best water for houseplants.
1.1 The Chlorine Myth and the Chloramine Reality
For generations, the standard advice for indoor gardeners was simple: ‘Fill your watering can and let it sit out for 24 hours.’ The logic was sound. Municipalities used chlorine (Cl2) to disinfect water. Chlorine is a volatile gas dissolved in liquid. Given enough surface area and time, it simply evaporates—or ‘off-gases’—into the atmosphere. By the next day, you had relatively chlorine-free water.
That advice is now dangerously outdated for most urban dwellers.
Water treatment plants faced a problem: chlorine evaporates too fast. It disappears before the water reaches the end of the distribution pipes, leaving the water vulnerable to re-contamination. To solve this, modern facilities switched to Chloramines—a chemical compound formed by binding chlorine with ammonia (NH₂Cl).
The chemical bond in chloramine is robust. It is designed not to break down. It does not evaporate. You can leave a bucket of chloraminated water sitting out until the next ice age, and it will still contain chloramine.
Why does this matter? Chloramine is a biocide. It kills bacteria. In the soil of a potted plant, your goal is to cultivate a rich microbiome of beneficial bacteria and mycorrhizal fungi that help the plant absorb nutrients. When you water with chloramine, you are essentially nuking that microbiome with every drink. Furthermore, chloramines can be directly cytotoxic to sensitive root hairs, burning them and reducing the plant’s ability to uptake water.
If you are relying on the ‘sit out’ method, you are likely dosing your soil with ammonia and chlorine every week. The only way to remove chloramine is through catalytic carbon filtration or chemical neutralization (like Vitamin C/Ascorbic acid or sodium thiosulfate—common aquarium water conditioners).
1.2 The Fluoride Menace (F–)
If Chloramine is a nuisance, Fluoride is a villain. Added to water supplies to prevent dental caries in humans, Fluoride (F–) is a cumulative poison for plants.
Here is the botanical reality: Plants do not have teeth. They have absolutely no biological requirement for fluoride. It is a non-essential element. However, plants are dumb pumps. They take up water through their roots via transpiration—the process where water evaporates from the leaves, creating a vacuum that pulls water up from the soil. Fluoride ions dissolve in water and ride this ‘transpiration stream’ straight up into the plant.
The Trap Mechanism: Once the water reaches the leaf, it evaporates through stomata (pores). But the fluoride? It doesn’t evaporate. It stays behind. It is left stranded at the very terminus of the journey: the tips and margins of the leaves.
Over time, this accumulation reaches toxic levels. It creates the classic ‘Necrotic Tip Burn’. You see this on your Spider Plants (Chlorophytum comosum), your Corn Plants (Dracaena fragrans), and your Prayer Plants (Calathea). You trim the brown tip off, thinking it’s dry air. Two weeks later, the brown tip returns. Why? Because the plant just pushed more fluoride into the new tip. You aren’t fighting humidity; you are fighting bio-accumulation.
Recent research has illuminated exactly how fluoride kills plant cells. It’s not just a chemical burn. Fluoride inhibits crucial enzymes like enolase (which powers glycolysis) and ATPase (which manages cellular energy). It effectively unplugs the cell’s battery. Furthermore, high fluoride concentrations trigger an ‘oxidative burst,’ generating Reactive Oxygen Species (ROS) like hydrogen peroxide that shred cell membranes from the inside out.
1.3 The Sodium Trap: The Water Softener Disaster
If you live in an area with ‘hard’ water—water rich in calcium and magnesium carbonates—you might have a water softener installed. These systems are great for your plumbing and your soap lather, but they are catastrophic for plants.
Water softeners work on the principle of ion exchange. They contain resin beads charged with sodium (Na+). As hard water flows over the beads, the calcium (Ca2+) and magnesium (Mg2+) stick to the resin, and sodium is released into the water to replace them.
You are literally swapping two essential plant nutrients (Calcium and Magnesium) for a plant toxin (Sodium).
The Osmotic Nightmare: Plants operate on osmosis. Water moves from areas of low salt concentration (the soil) to areas of high salt concentration (the root cells). When you water with softened water, you are loading the soil with sodium. Eventually, the salt concentration in the soil becomes higher than in the root.
The physics flips. The soil starts sucking water out of the root. This is ‘physiological drought.’ You can water your plant every single day, keeping the soil soaking wet, and the plant will wilt and die of dehydration because the salt won’t let it drink.
The ‘Street-Smart’ advice here is absolute: Never, ever water plants with softened water. Use the bypass tap, usually found in the garage or garden hose spigot.
1.4 The ‘Hardness’ Dilemma: pH and Lockout
Hard water itself—unsoftened—is a mixed bag. It contains high levels of Calcium Carbonate (CaCO₃). While Calcium is good, the Carbonate part acts as a powerful pH buffer. It keeps the pH of the water high (alkaline), often above 7.5 or 8.0.
Most tropical houseplants evolve in acidic rainforest soils (pH 5.5–6.5). When you constantly water with alkaline tap water, you slowly raise the pH of the potting mix.
- The Lockout: As soil pH climbs above 7.0, essential micronutrients like Iron (Fe) and Manganese (Mn) become chemically insoluble. They turn into solids that the roots cannot absorb.
- The Symptom: Interveinal Chlorosis. The leaves turn yellow while the veins stay green. You might think the plant needs fertilizer, so you feed it. But the plant can’t eat the food because the pH door is locked. More fertilizer just adds more salts, exacerbating the problem.
Part2: The Fluoride Menace — A Deep Dive into Phytotoxicity

To understand why we are searching for the best water for houseplants, we have to respect the damage Fluoride does. It is insidious because it is invisible until the damage is done.
2.1 The ‘Canaries in the Coal Mine’
Not all plants handle Fluoride the same way. Research identifies distinct categories of sensitivity.
Table 1: Fluoride Sensitivity in Common Houseplants
| Sensitivity Level | Plant Genera | Symptoms of Toxicity |
|---|---|---|
| High Sensitivity | Dracaena (Corn Plant, Dragon Tree), Chlorophytum (Spider Plant), Spathiphyllum (Peace Lily), Calathea, Maranta, Stromanthe | Rapid necrosis of leaf tips; distinct yellow band (chlorosis) separating dead and living tissue; leaf elongation inhibition. |
| Moderate Sensitivity | Monstera, Philodendron, Epipremnum (Pothos), Yucca | Slower accumulation; marginal browning usually only on older leaves; general lack of vigor. |
| Accumulators (Tolerant) | Camellia sinensis (Tea), Ficus elastica (Rubber Plant) | Can store high levels of F without visible symptoms initially, but eventually suffer photosynthetic decline. |
If you own a ‘High Sensitivity’ plant, tap water is essentially a slow death sentence. These plants evolved in environments where water was mineral-free rainwater. They lack the genetic machinery to sequester or excrete fluoride.
2.2 The Cellular Mechanism of Death
Recent studies, particularly from 2021–2025, have utilized advanced imaging to watch fluoride move through plant tissues. When Fluoride enters the root, it bypasses the Casparian Strip—the root’s natural bouncer. The Casparian strip is supposed to filter out unwanted ions, but Fluoride is sneaky. It is small and highly electronegative. It slips through the apoplastic pathway (the space between cell walls) and enters the xylem.
Once in the leaf tip, Fluoride disrupts photosystem II. This is the engine of photosynthesis. It literally stops the plant from turning light into energy. This causes the chlorosis (yellowing).
Simultaneously, the accumulation triggers the cell to panic. The cell produces Reactive Oxygen Species (ROS)—specifically Superoxide (O₂-) and Hydrogen Peroxide (H₂O₂). These are oxidative radicals. They act like biological rust, stripping electrons from cell membranes (lipid peroxidation) until the cell wall collapses.
- Result: The tissue turns brown, papery, and dead. This is irreversible.
2.3 The ‘Tea Plant’ Proxy
Much of our deep understanding of this comes from the tea industry. Tea plants (Camellia sinensis) are hyper-accumulators of fluoride. We drink tea, so scientists care a lot about how much fluoride ends up in the leaves.
A landmark 2024 study investigated how to stop tea plants from sucking up so much fluoride. They found that applying Calcium (Ca) and Magnesium (Mg) changed the structure of the cell wall, making it harder for fluoride to stick. This is a crucial clue for our houseplants: Nutrition affects Toxicity.
Part3: The Silicon Revolution — The Missing Link

For years, Silicon (Si) was the ‘forgotten nutrient.’ It wasn’t considered ‘essential’ because plants can technically survive without it—kind of like how a human can technically survive living in a bubble. You are alive, but you aren’t thriving, and the first germ that touches you will kill you. Even if you have the best water for houseplants, a lack of silicon can still leave them vulnerable.
In the wild, plants grow in soil. Soil is mostly silica (sand, clay, quartz). As rocks weather, they release trace amounts of Monosilicic Acid (H₄SiO₄), which plants drink up. But look at your potting mix. It’s peat moss, coco coir, and perlite.
- Peat/Coco: Organic matter. Zero silicon.
- Perlite: Volcanic glass. Technically silica (SiO₂), but chemically insoluble. It’s locked in a glass matrix. The plant can’t access it any more than you can eat a windowpane to get calcium.
The Reality: Indoor plants are growing in a Silicon Vacuum. They are structurally deficient.
3.1 The ‘Armor’ Hypothesis
Research published between 2020 and 2025 has cemented Silicon’s role as a ‘quasi-essential’ element for stress resistance. When a plant has access to soluble Silicon, it absorbs it and deposits it in two key places:
- The Cell Walls (Phytoliths): Silicon polymerizes inside the cell walls, forming ‘biogenic opal’ (SiO₂. nH₂O). This acts like rebar in concrete. It physically hardens the plant tissue.
- Effect: The leaves become tougher. They are harder for pests to chew (literally wearing down the mandibles of caterpillars). They lose less water to evaporation.
- The Root Endodermis: This is the game-changer for our water quality problem. Silicon reinforces the Casparian strip in the roots.
3.2 The Mechanism of Fluoride Blockade
Here is the exciting part. A 2025 study on wheat demonstrated that Silicon supplementation actively reduces Fluoride toxicity through three distinct mechanisms:
- Exclusion (The Bouncer): Silicon promotes the deposition of Suberin in the roots. Suberin is a waxy, corky substance. By thickening the suberin layer, the root becomes less permeable to passive ion flow. The ‘bouncer’ at the door gets bigger muscles. The study showed that Si-treated plants took up significantly less Fluoride than untreated ones, even when swimming in the same toxic water.
- Antioxidant Upregulation (The Cleanup Crew): Remember the ROS (oxidative rust) that Fluoride causes? Silicon acts as a signaling molecule. It tells the plant, ‘We are under attack, arm the defenses!’ The plant responds by producing massive amounts of antioxidant enzymes: Superoxide Dismutase (SOD) and Catalase (CAT). These enzymes hunt down the ROS radicals and neutralize them before they can destroy the cell membrane. The Silicon doesn’t fight the toxin directly; it trains the plant to fight it.
- Transpiration Reduction: By thickening the leaf cuticle with silica, the plant transpires less. Since Fluoride accumulation is driven by the volume of water moving through the plant, slowing down the water flow (without stressing the plant) reduces the total load of toxins accumulating in the tips.
This is the ‘Street-Smart’ translation: Silicon turns your delicate Calathea into a tank. It armors the roots against toxin entry and arms the cells against oxidative damage.
Part4: The Silicon Marketplace — Avoiding Snake Oil

Now that you are hyped on Silicon, you might rush to Amazon and buy the first bottle that says ‘Silica.’ Stop. The fertilizer industry is rife with confusion here. Not all Silicon is created equal.
4.1 The Three Forms of Silicon
Understanding these three forms is the difference between wasting money and saving your plants.
Table 2: Silicon Sources Comparison
| Form | Chemical Name | Bioavailability | pH Effect | Stability | The ‘Street’ Verdict |
|---|---|---|---|---|---|
| Sand / Perlite / DE | Silicon Dioxide (SiO₂) | < 1% (Insoluble) | Neutral | High | Useless for immediate uptake. Good for soil structure, but don’t count on it for nutrition. |
| Potassium Silicate | K₂SiO₃ | 50–70% (Needs conversion) | High Alkaline (Raises pH) | High | The Standard. Cheap, effective, but messy. Requires pH balancing. |
| Monosilicic Acid | H₄SiO₄ (MSA) | 100% (Plant Available) | Neutral | Low (Polymerizes easily) | The Ferrari. Instant uptake, no pH mess, but very expensive. |
4.2 Potassium Silicate: The Workhorse
This is what you find in bottles like ‘Pro-TeKt,’ ‘Armor Si,’ or ‘Silica Blast.’ It is cheap and effective. However, it has a high pH (often 10+).
- The Reaction: When you mix it into water, it needs time to hydrolyze (break down) from long silicate chains into the monomer form (H₄SiO₄) that plants can drink.
- The Risk: If you mix it wrong, it creates Nutrient Lockout.
The Mixing Protocol (The ‘Bucket Chemistry’ Rule): You cannot just dump Potassium Silicate into your watering can with your other fertilizers. If silicate meets concentrated Calcium (from CalMag) or Magnesium, it instantly reacts to form Calcium Silicate. Calcium Silicate is sand. It is insoluble. You will see the water turn cloudy white. That cloud is your expensive nutrients precipitating out of the solution and becoming useless dirt.
The Correct Protocol:
- Water First: Fill your container with clean water.
- Silica Second: Add the Potassium Silicate. Stir vigorously. Wait 10–20 minutes. This waiting period is crucial. It allows the silicate to disperse and hydrolyze.
- pH Check (Optional): The pH will spike. If it’s extremely high, you might lower it slightly, but usually…
- Fertilizer Third: Add your liquid fertilizer. The acidity of most fertilizers will naturally pull the pH back down, neutralizing the alkalinity of the silica.
- CalMag Last: Add Calcium/Magnesium supplements last.
4.3 Monosilicic Acid (MSA): The ‘Gold’ Standard
Products like ‘Dune,’ ‘Power Si,’ or ‘Alchemist’ use stabilized Monosilicic Acid.
- Why use it: It is instantly available. It doesn’t spike pH. You can use much less of it (droplets vs. teaspoons).
- The Catch: It is chemically unstable. It wants to turn back into sand. Manufacturers use proprietary stabilizers (often alcohol or acids) to keep it liquid. This makes it expensive—often $50–$100 a bottle.
- Is it worth it? For a rare variegated Monstera that costs $500? Yes. For a Spider Plant? Probably not.
Part5: Wollastonite — The Lazy Expert’s Secret Weapon

What if I told you there is a way to get the benefits of Calcium and Silicon without measuring liquids, checking pH, or worrying about precipitation order? Enter Wollastonite.
This is the deep cut from the research snippets. Wollastonite is a naturally occurring mineral: Calcium Silicate (CaSiO₃). Wait, didn’t I just say Calcium Silicate is insoluble sand? In the context of mixing liquid nutrients, yes, precipitating calcium silicate is bad because it falls out of solution. But in the context of Soil Amendment, Calcium Silicate is magic. It slowly transforms ordinary moisture into the best water for houseplants right inside the pot.
Ref:
https://doi.org/10.1021/acsomega.8b02477
https://doi.org/10.3389/fpls.2022.929457
https://doi.org/10.1007/s10705-025-10437-1
5.1 The Slow-Release Mechanism
Wollastonite is a mineral that weathers (breaks down) in the soil. But unlike quartz sand which takes geological eons to break down, Wollastonite weathers relatively quickly on a biological timescale—weeks to months. As it breaks down, it releases two things into the root zone:
- Calcium Ions ()
- Silicate Ions ()
It provides a steady, drip-feed supply of the exact two elements we need to fight Fluoride toxicity.
5.2 The Carbon Sequestration Bonus
Here is the ‘surprise’ factor mentioned in the prompt requirements. Using Wollastonite actually fights climate change in your living room. Enhanced Weathering: When Wollastonite breaks down, the calcium reacts with Carbon Dioxide (CO₂) in the soil (produced by root respiration) to form Calcium Carbonate (CaCO₃).
This process literally traps atmospheric CO₂ and turns it into stable rock in your pot. Research shows that amending soil with Wollastonite can sequester significant carbon while boosting plant biomass by up to 40%.
Ref: https://doi.org/10.3389/fpls.2022.929457
5.3 The ‘Set It and Forget It’ Protocol
This is the ultimate hack for the casual hobbyist.
- Dosage: Add about 1 tablespoon of Wollastonite powder per gallon of soil when repotting. Or, top-dress your existing plants with a dusting of it and water it in.
- Effect: It buffers the soil pH (keeping it from getting too acidic, which releases more fluoride). It supplies Calcium. It supplies Silicon.
- Where to get it: It’s often sold as a soil amendment in agricultural stores, or sometimes rebranded in the cannabis growing market. It is cheap, shelf-stable, and impossible to ‘overdose’ because it’s a rock.
Part6: Finding the Best Water for Houseplants — A Hierarchy

We have covered the chemistry. Now let’s look at the actual water sources available to you and rank them based on our new understanding of what constitutes the best water for houseplants.
6.1 Rainwater: The Gold Standard (With Caveats)
Rainwater is what plants evolved to drink. It is naturally soft, slightly acidic (pH 5.5–6.0 due to dissolved CO₂), and contains dissolved oxygen and trace nitrates (nitrogen from lightning storms). It is widely considered the best water for houseplants.
- Pros: Free. Perfect pH. No fluoride. No chlorine.
- Cons: Storage is a pain. Stagnant rainwater breeds mosquitoes and anaerobic bacteria (the ‘rotten egg’ smell). In heavy urban areas, the ‘first flush’ of rain can be polluted with smog particulates and heavy metals.
- The Fix: If you collect rain, use it fresh. If storing, keep it in a dark, sealed container to prevent algae growth.
6.2 Reverse Osmosis (RO) and Distilled Water: The Blank Slate
This is water stripped of everything. 0 ppm (Parts Per Million) of dissolved solids.
- Pros: Guaranteed free of Fluoride, Sodium, and Chlorine. It is the safest baseline for sensitive plants like Calathea.
- Cons: It is ‘hungry’ water. Because it has no minerals, it is hypotonic. If you water with pure RO water, it can actually leach nutrients out of the plant roots in an attempt to balance equilibrium.
- The Fix: You must remineralize RO water. You cannot use it straight forever. You need to add a ‘CalMag’ supplement (Calcium/Magnesium) to bring the EC (Electrical Conductivity) up to a safe level (around 150–200 ppm) before watering.
6.3 Tap Water: The ‘Manageable Risk’
Most of us are stuck with this.
- Pros: Convenient. Contains Calcium and Magnesium (if hard).
- Cons: The ‘Big Three’ toxins (Chlorine, Fluoride, Sodium).
- The Protocol:
- Dechlorinate: Use an aquarium water conditioner (like Seachem Prime or generic Sodium Thiosulfate). Two drops per gallon instantly neutralizes Chlorine and Chloramines. It’s cheap and effective. Stop waiting 24 hours.
- Mitigate Fluoride: You can’t filter it out easily. So, you must use the Silicon/Calcium defense. If you use tap water, you must be using a Silicon supplement or Wollastonite in your soil. This is your insurance policy.
- Check pH: Get a cheap pH test kit (drops or strips). If your tap water is pH 8.0+, use a ‘pH Down’ product (phosphoric acid) to bring it to 6.5. Your plants will thank you.
6.4 Greywater: The ‘Do Not Touch’ Zone
With sustainability in mind, people ask about using bathwater or dishwater (greywater). Research is clear: Don’t use it for container houseplants.
- Why: Greywater is full of surfactants (soaps) which are chemically salts (Sodium). They drastically raise alkalinity (pH 10+). They coat roots and prevent water uptake.
- Exception: Outdoor gardens with robust soil microbiomes can handle it. A potted Ficus cannot.
6.5 Aquarium Water: The Secret Superfood
If you have a freshwater fish tank, the water you remove during a water change is ‘Liquid Gold.’ It rivals rainwater as the best water for houseplants.
- Why: It is dechlorinated. It is room temperature. It is rich in Nitrates (NO₃) and Phosphates (PO₄) from fish waste—the exact macro-nutrients plants need.
- Caveat: Do not use if you have added salt to your aquarium or if you have recently medicated the fish.
Part7: Plant-Specific Battle Plans

Let’s translate this entire report into actionable advice for specific plant families. Choosing the best water for houseplants often depends on the specific variety you are growing.
7.1 The ‘Fluoride Canaries’ (Dracaena, Spider Plant, Peace Lily)
These are the plants that get brown tips if you even look at them wrong.
- Water: Strictly Rainwater, RO, or Distilled. They have zero tolerance for fluoride.
- Soil: Must include Wollastonite or a Silicon amendment.
- pH: Keep soil pH between 6.0 and 6.5. If it drops below 6.0, any trace fluoride in the soil becomes hyper-available. Avoid heavy peat moss (which acidifies); add extra pumice or charcoal.
7.2 The ‘Divas’ (Calathea, Maranta, Ferns)
These guys suffer from ‘Crispy Edge Syndrome.’
- The Cause: Often misdiagnosed as humidity issues. It is usually osmotic stress from salts (sodium) or chloramines damaging the fine root hairs.
- The Fix: Use RO/Distilled water. If you use tap, you must dechlorinate chemically.
- Silicon Trick: Foliar spray! You can buy Monosilicic Acid sprays (or mix your own weak solution). Spraying the leaves deposits silicon directly into the cuticle, hardening the leaf and reducing water loss. This makes them much more tolerant of dry indoor air.
7.3 The ‘Investors’ (Monstera Albo, Philodendron var.)
You spent $200 on a cutting. The white parts are turning brown (melting).
- The Science: The white tissue lacks chlorophyll. It produces no energy. It is structurally weak parasite tissue relying on the green parts. It is the first to die when the plant is stressed.
- The Fix: Silicon supplementation is critical here. By reinforcing the cell walls of the white tissue with silica, you prevent the cell collapse that leads to browning. ‘Silica is the glue that holds the variegation together’ is the street-smart mantra here.
7.4 The ‘Succulents & Cacti’
- Water: Tap water is usually fine. They are tough.
- Hardness: They actually appreciate the Calcium in hard water.
- Warning: They are very susceptible to root rot if the water has high pathogens. Do not use greywater.
Part8: Conclusion: The Search for the Best Water for Houseplants Ends Here
The days of simply filling a watering can and hoping for the best are over. We know too much now. The ‘Black Thumb’ is often just a symptom of a chemical mismatch between municipal engineering and biological evolution.
We have learned that:
- Water isn’t just wet. It’s a chemical delivery system.
- Fluoride and Chloramine are invisible assassins. They work slowly, attacking roots and leaf tips.
- Silicon is the armor. It is the missing piece of the puzzle that fortifies plants against our imperfect water.
- Wollastonite is the cheat code. A simple rock dust that fixes pH, adds Calcium, and supplies Silicon.
The ‘Best Water for Houseplants’ Recipe: If you want the absolute best results for your indoor jungle, here is the formula:
- Source: Reverse Osmosis Water.
- Remineralization: A liquid ‘CalMag’ supplement to 150 ppm.
- Fortification: Monosilicic Acid (0.5 ml/Gallon).
- pH: Adjusted to 6.0.
The ‘Good Enough’ (Street-Smart) Recipe:
- Source: Tap Water.
- Treatment: Aquarium Dechlorinator (2 drops/Gallon).
- Fortification: Top dress your soil with Wollastonite powder once a year.
- Observation: Watch the tips. If they brown, increase the Silicon.
You are now armed with the botany, biology, and chemistry to look at that glass of water differently. Stop poisoning your plants. Start armoring them. You finally know the truth about the best water for houseplants.
Appendix: Data Tables & Quick References
Table 3: The ‘Tip Burn’ Troubleshooting Guide
| Symptom | Probable Cause | The Fix |
|---|---|---|
| Browning at very tip, yellow ‘halo’ | Fluoride Toxicity | Switch to RO water; Add Silicon. Check pH (aim for >6.0). |
| Crispy edges all around leaf | Salt Stress / Underwatering | Flush soil with distilled water to remove salt buildup. Stop using softened water. |
| Yellowing between veins (Green veins) | Nutrient Lockout (pH too high) | Water is too alkaline (Hard). Use pH Down or switch to Rainwater. |
| Yellowing of lower leaves | Nitrogen Deficiency or Mobile Nutrient Scavenging | Plant is eating itself. Increase fertilizer. |
| Mushy brown spots | Bacterial/Fungal Infection (Root Rot) | Check roots. Stop watering. Increase airflow. |
Table 4: Water Quality Hierarchy
| Grade | Water Type | Notes |
|---|---|---|
| S-Tier | Rainwater, Aquarium Water | Biologically active, chemically gentle. The gold standard. |
| A-Tier | RO/Distilled + CalMag | The ‘Control Freak’ option. Perfect but requires effort. |
| B-Tier | Tap Water + Dechlorinator + Silicon | The ‘Realist’ option. Workable for most plants if managed well. |
| F-Tier | Softened Water, Untreated Greywater | Toxic. Do not use. |

Leave a Reply