Summary
- Modern soilless potting mixes deprive indoor plants of Silicon, a critical element wild plants use to build structural “glass armor” (biosilicification) against environmental threats.
- Supplementing with Silicon fortifies cell walls and forms microscopic physical barriers, drastically increasing resistance to drought, pests (like thrips and mites), and fungal diseases.
- To reverse “soft” growth, hobbyists should incorporate Potassium Silicate (for immediate uptake) or Diatomaceous Earth (for slow release) into their routine, following strict mixing protocols to avoid nutrient lockout.
Key Takeaways
- The “Glass” Defense: Silicon polymerizes into opal-like structures (phytoliths) inside leaves, which physically wear down the mandibles of chewing pests and block the stylets of sucking insects.
- Drought & Stress Buffer: Silicon reinforces the leaf cuticle to reduce water loss by up to 30% and helps plants manage salt buildup from tap water and fertilizers by blocking sodium uptake.
- Essential for Aroids: Popular houseplants like Monstera and Philodendron are active silicon accumulators that rely on it for stem rigidity and leaf toughness; without it, they are structurally “flabby” compared to their wild counterparts.
- Critical Mixing Rule: When using Potassium Silicate (the most effective source), you must add it to the water first, lower the pH (as it is highly alkaline), and then add your fertilizer to prevent nutrients from precipitating into “cement”.
Introduction: The Great “Sterile Soil” Swindle and Missing Plant Nutrients in Soil
Your expensive, rare houseplants are probably biological wimps. You’ve followed every influencer’s advice—chunky soil, high-end fertilizers, and 24/7 humidifiers—yet they still wilt the moment you miss a watering or a single bug hitches a ride. In the wild, these same species are indestructible tanks that survive monsoons and pests without a drop of distilled water.
The difference isn’t just “nature”—it’s the chemistry of the dirt. Most hobbyists feed their plants a basic NPK multivitamin while completely ignoring the structural building blocks they find in the earth. Specifically, you’re missing Silicon (Si).
While old textbooks labeled Silicon as “non-essential,” recent research from the last five years proves that for Aroids and ferns, Silicon is the difference between a plant on life support and a biological fortress. It’s time to stop treating your collection like delicate glass ornaments and start helping them build themselves out of actual glass. Here is the science of why your current potting mix is sabotaging your plants and how Silicon-driven biochemistry creates “Glass Plants” that can actually defend themselves.
Part 1: The “Non-Essential” Lie About Plant Nutrients in Soil
The Historical Blind Spot
To understand why your potting mix is failing you, you have to understand the history of “essential nutrients.” In 1939, two scientists named Arnon and Stout laid down the law of what makes an element essential. They decided that an element is only essential if the plant physically cannot complete its life cycle (i.e., make seeds) without it. Nitrogen fits this. Without Nitrogen, no DNA, no proteins, plant dies. Magnesium fits this. Without Magnesium, no chlorophyll, no photosynthesis, plant dies.
Silicon? Silicon is tricky. It is the second most abundant element in the Earth’s crust, making up about 28.8% of the ground beneath our feet.
https://pmc.ncbi.nlm.nih.gov/articles/PMC9182727
Because it is everywhere in nature, plants evolved in a soup of dissolved silica. They didn’t need to develop a strict “death switch” for when it was missing, because in the wild, it was never missing. It’s like oxygen for humans; we don’t have a backup plan for a lack of oxygen because it’s always there.
But then humans invented “soilless culture.” We started growing plants in peat moss (decayed sphagnum from bogs), coconut coir (husk fiber), and perlite (volcanic popcorn). These substrates are fantastic for air porosity, preventing root rot, and convenience. But chemically? They are barren. Peat moss is organic matter; it has almost zero mineral content. Coconut coir is lignin and cellulose; it has no minerals. Perlite is technically volcanic glass (SiO2), but it is insoluble. It’s like trying to get your daily iron intake by licking a steel beam. It’s there, but your body can’t access it.
https://www.mdpi.com/2311-7524/7/9/263
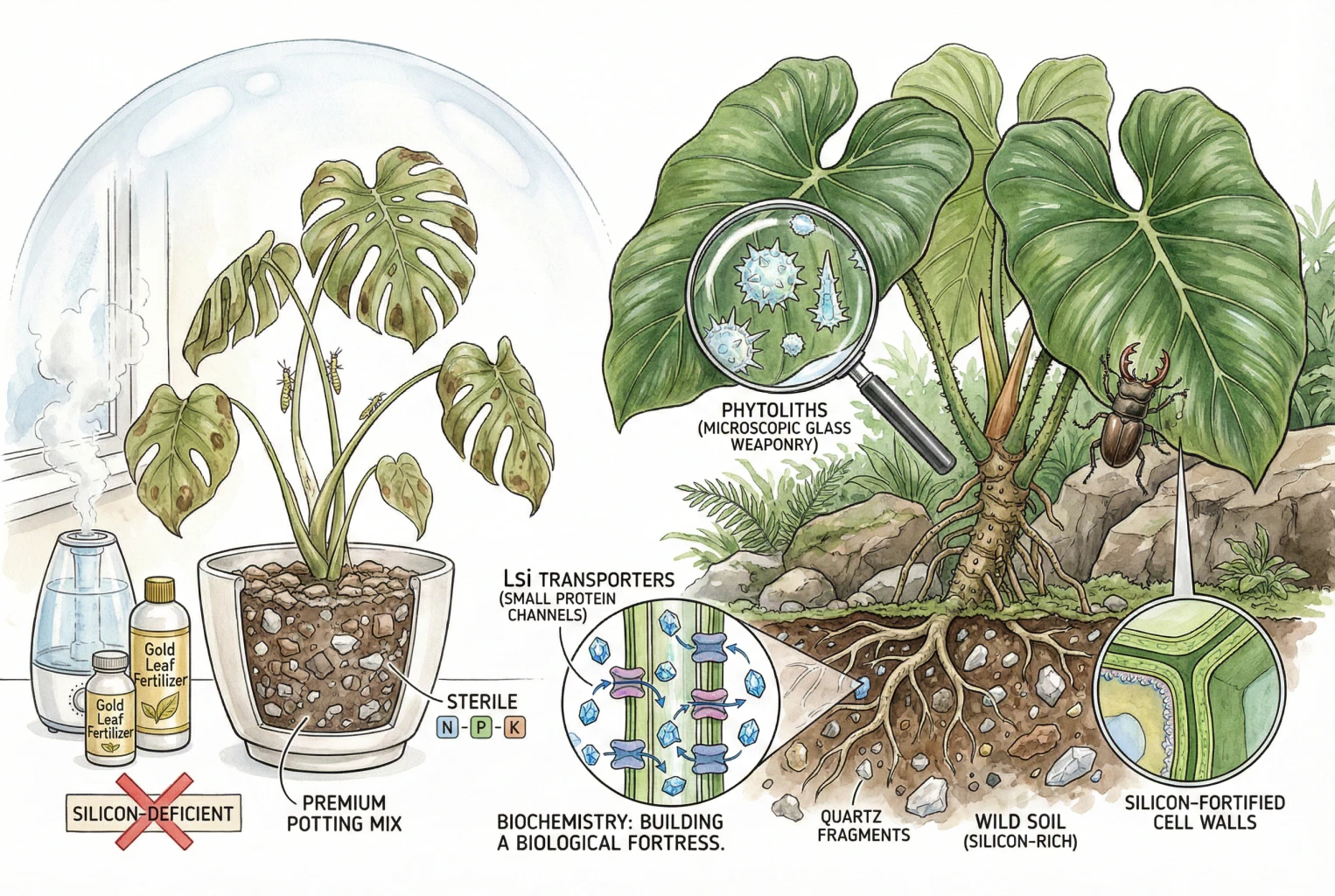
So, we took plants that evolved to root into mineral-rich earth, dissolving rocks with their root acids to absorb silica, and we put them in plastic pots filled with fluffy, nutrient-void fiber. We effectively stripped them of their armor. We created “flabby” plants. They lack mechanical stiffness. They droop. Their cell walls are thinner. And when we read the back of our fertilizer bottles, we see N-P-K, maybe some Calcium or Iron, but Silicon is almost never there.
The Transformation: Biosilicification
When you reintroduce soluble silicon to a plant that has been starved of it, the transformation is nothing short of alchemical. The plant takes up the silicon and undergoes a process called biosilicification. It turns a liquid nutrient into solid stone inside its own body.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12474856
This isn’t a metaphor. The plant absorbs silicon as Monosilicic Acid (H4SiO4), which is a water-soluble molecule. It travels up the roots, through the stems, and into the leaves. Once it hits the leaves, the water evaporates (transpiration). As the water leaves, the silicon stays behind. The concentration rises until it hits a saturation point and polymerizes. It turns from a liquid acid into a solid gel (SiO2⋅nH2O), effectively amorphous glass.
https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2017.00411/full
This “biogenic opal” is deposited in specific locations that the plant needs to reinforce. It lays it down in the cell walls, cross-linking with hemicellulose to create a structure that is both rigid and elastic. It lays it down just under the waxy skin of the leaf (the cuticle), creating a double-layer shield of wax and glass. It even builds specific structures called Phytoliths—microscopic shapes like dumbbells or saddles—that float in the cell and act as internal support struts and anti-herbivore weaponry.
The Mechanism of Uptake: Why Some Plants Are “Eaters” and Others Are “Drinkers”
Not all plants are created equal in their hunger for glass. This is where the botany gets specific, and it explains why your Aroids and grasses respond so well while your cactus might not care as much.
Research has identified specific protein channels in the roots dedicated to grabbing silicon from the soil. These are named Lsi1 (influx) and Lsi2 (efflux) transporters.
https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2021.697592/full
The Lsi1 transporter sits on the outside of the root cell. Its job is to grab Silicic Acid from the soil water and pull it into the root. The Lsi2 transporter sits on the inner side, pumping that silicon into the plant’s vascular system (the xylem) so it can be shot up to the leaves.
Plants are categorized based on how many of these transporters they have:
- Active Accumulators: These plants have high-powered Lsi1 and Lsi2 pumps. They actively hunt for silicon and concentrate it in their tissues at levels higher than the soil solution. This group includes Rice, Sugarcane, Bamboo, and—crucially for us—many members of the Araceae (Aroids) and Commelinaceae (Tradescantia) families. These plants crave silicon. If you don’t give it to them, they suffer structural weakness.
- Passive Accumulators: These plants take up silicon roughly in proportion to how much water they drink. They have some channels, but they aren’t pumping it aggressively. Many broadleaf dicots (like Ficus or Roses) fall here. They still benefit massively, but they don’t hoard it like the accumulators do.
- Excluders: These plants actively reject silicon. This is rare, mostly seen in some specialized ecological niches.
For the indoor gardener, this means your Monstera, Philodendron, Alocasia, and Anthurium are likely Active Accumulators or high-level Passive Accumulators. They evolved to use silicon as a primary structural component. Depriving them of it is like building a skyscraper out of wood instead of steel. It might stand up for a while, but the first storm is going to cause problems.
https://www.mdpi.com/2223-7747/9/12/1779
Part 2: The Abiotic Shield – Surviving Your Neglect
We need to address the elephant in the room: human error. We all kill plants. We don’t mean to, but life gets in the way. We forget to water for a week. We leave the blinds open on a scorchingly hot day. We mix the fertilizer too strong because we think “more is better.”
This is called Abiotic Stress. It is stress caused by non-living factors: drought, heat, salt, cold. And this is where Silicon shines. It acts as a buffer for your mistakes. It widens the margin of error between “oops” and “dead plant.”
1. Drought Tolerance: The “Waterproofing” Upgrade
Drought is the number one killer of house plants. You go on vacation, or you just get busy. The soil dries out. The roots lose contact with water. The turgor pressure (the internal water pressure that holds the plant up) drops. The plant wilts. If it stays wilted too long, the cells collapse and die.
Silicon acts as a waterproofing upgrade for your plant’s hydration system. Research on crops like Sorghum and Maize—which are physiological cousins to many of our decorative monocots—shows that Si application significantly mitigates the damage caused by water deficiency.
There are three distinct mechanisms at play here, and they are fascinating.
Mechanism A: The Silica-Cuticle Double Layer A huge amount of water is lost from a plant not through the breathing pores (stomata), but simply by evaporating through the skin of the leaf. This is called cuticular transpiration. It’s wasteful. It’s like a leaky pipe. When a plant has access to Silicon, it deposits that layer of glass right underneath the waxy cuticle. This creates a significantly more impermeable barrier. It seals the leaks. Research indicates this can reduce passive water loss by up to 30%.
Think about what that means for your Calathea. Calatheas are notorious for curling their leaves the moment the humidity drops or the soil dries. That curling is a panic reaction to stop water loss. A Calathea fortified with silicon has a better seal. It loses less water to the dry apartment air. It doesn’t panic as quickly. It stays open and happy for days longer than its unfortified sibling.
Mechanism B: Stomatal Control The stomata are the pores that let CO2 in and water out. In a drought, the plant needs to close them to save water. Silicon seems to make the stomata more sensitive and efficient. It allows the plant to snap them shut faster when conditions get dry, preventing catastrophic water loss before it happens.
https://pubs.acs.org/doi/10.1021/acsomega.0c03820
Mechanism C: Aquaporin Regulation This is the deep science. Aquaporins are microscopic channels in the cell membranes of the roots that specifically allow water to pass through. They are the floodgates. Recent studies suggest that Silicon upregulates the gene expression of these aquaporins. It essentially tells the root system to open more floodgates and scavenge every last molecule of water from the soil. It increases root hydraulic conductance—the ease with which water moves from the dirt into the vein. So, when your soil is barely damp and a normal plant would be wilting, the Si-treated plant is still managing to pull moisture out and keep its leaves stiff.
2. Salt Stress and The “Crispy Tip” Syndrome
If you have ever grown a Dracaena, a Spider Plant, or a Calathea, you know the scourge of the “crispy brown tip.” You trim it off, and it comes back. The internet tells you it’s “chemicals in the water” or “fluoride.”
While fluoride is a factor, the main culprit is often Salt Stress. This comes from two sources:
- Tap Water: If you live in an area with hard water, it is full of Sodium, Chlorides, and Carbonates.
- Fertilizer Buildup: Every time you feed your plant, you are adding salts (Nitrates, Phosphates). If you don’t flush the pot perfectly every time, these salts accumulate.
Salt is deadly because it competes with water. It creates osmotic pressure that pulls water out of the roots. It also rushes into the plant and burns the leaf margins, which is the furthest point from the roots and where the salts get dumped.
Silicon is the antidote to salt stress. Recent studies (2020–2025) have highlighted its role in filtering out these toxins.
https://www.mdpi.com/2073-4395/9/8/460
The “Bypass Block” Mechanism: In a root, water can travel two ways: through the cells (Symplastic) or through the gaps between the cell walls (Apoplastic). The Apoplastic route is dangerous because it bypasses the cell’s filtration systems. It’s a back door for sodium to sneak in. Research on rice (the model organism for monocots) shows that Silicon deposits in the Endodermis (the inner skin of the root) and physically blocks this “apoplastic bypass” flow. It acts like caulk, sealing up the gaps between the cells. It forces all the water to go through the cell membranes, where the plant’s biological bouncers can kick the sodium out.
Ion Compartmentalization: Even if sodium gets in, Silicon helps the plant deal with it. It helps the plant sequester the sodium in the vacuole (the cell’s trash can) so it doesn’t float around damaging the cytoplasm. Simultaneously, it helps the plant keep taking up Potassium (K), which is chemically similar to Sodium but essential for life. Usually, Sodium blocks Potassium uptake. Silicon restores the balance.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11375724
Real Life Application: If you can’t afford a reverse osmosis system and are stuck watering your Calathea with tap water, Silicon is your safety net. It won’t fix the water, but it will armor the plant against the salts in it. It reduces the crispiness. It keeps the edges green.
3. Metal Toxicity: The “Heavy Metal Jacket”
While most of us aren’t gardening on toxic waste dumps, metal toxicity is a real issue in potting mixes. If the pH of your peat moss drops too low (becomes too acidic), naturally occurring metals like Manganese (Mn) and Aluminum (Al) become highly soluble. They flood the plant and cause toxicity, often showing up as weird crinkling or necrotic spots on leaves.
Silicon has a high affinity for binding with heavy metals. In the cell wall, it acts like a magnet, binding with Manganese or Aluminum and locking them in place. It immobilizes them. It essentially puts the heavy metals in a “straightjacket” so they can’t interfere with the plant’s enzymes.
A recent study on Chilli plants showed that Si reduced the uptake of Cadmium and Lead, protecting the photosynthetic machinery from damage.
https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1590148/full
While you hopefully don’t have lead in your living room, this mechanism applies to the micronutrient spikes that happen when we mess up our fertilizer dosing. It smoothens out the peaks of toxicity.
Part 3: The Biotic Shield – War Against Pests

This is where Silicon goes from “nice to have” to “essential warfare.” If you have been in the hobby long enough, you have battled The Big Three: Thrips, Spider Mites, and Mealybugs.
We usually fight these with chemical warfare—Neem Oil, Spinosad, Systemic Granules. These work, but they are harsh. They smell. They can burn leaves. And pests build resistance to them. You can’t evolve resistance to a wall of glass. Silicon offers a mechanical and chemical alternative that is evolution-proof.
1. The Physical Barrier: “Chewing on Sandpaper”
When Si polymerizes in the leaf, it forms those Phytoliths we mentioned earlier. These are microscopic, hard, opal structures. On the Mohs scale of mineral hardness, opal is about a 5.5 to 6.5. The mandibles (jaws) of most insects are made of chitin, which is much softer, around a 3.
The Mechanism vs. Chewing Pests (Caterpillars, Beetles): Imagine you are a caterpillar. You land on a juicy Monstera leaf. You take a bite. If that leaf is fortified with Silicon, you aren’t just chewing on salad; you are chewing on a salad mixed with sand and ground glass. Research shows that larvae fed on high-Si diets suffer from severe mandibular wear. Their teeth literally get ground down.
It hurts to eat. They expend massive amounts of energy trying to chew, and they get less nutrition out of it. Their growth rates plummet. Many starve to death or grow up to be stunted, weak adults that lay fewer eggs.
The Mechanism vs. Sucking Pests (Thrips, Mites, Aphids): These guys don’t chew; they pierce. They use a stylet (a biological needle) to punch into individual cells and suck out the juice.
- Thrips: They utilize a “slash and suck” technique. They rasp the surface of the leaf to break it open. A high-Si epidermis is tougher to rasp. It’s like trying to scratch through Kevlar with a plastic fork.
- Spider Mites: They pierce cells directly. If the cell wall is reinforced with Si, it is mechanically harder to puncture. The result is that the pests can still feed, but it is inefficient. They have to work harder for every meal. A population of spider mites on a Si-treated plant explodes much slower than on a soft, untreated plant. It buys you time to catch the infestation before it destroys the plant.
2. The Chemical Priming: “Calling the Cavalry”
This is the most exciting finding in recent years (2020–2024). We used to think Silicon was just a passive brick wall. We now know it acts as an active alarm system.
The Jasmonic Acid Pathway: Plants have an immune system. When they are attacked, they produce defense hormones, primarily Jasmonic Acid (JA). This hormone tells the plant to start producing toxins and bad-tasting compounds to drive the bugs away. In a normal, Si-starved plant, there is a “lag time.” The bug bites, and it takes a while for the JA levels to spike. It’s a slow reaction. Research suggests that soluble Si “primes” this pathway.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4904004
In a Si-treated plant, the defense machinery is on a hair-trigger. When a pest bites, the JA response spikes faster and higher. It’s the difference between a security guard who is asleep at the desk and one who is watching the monitors with his hand on the alarm button.
Tri-Trophic Interactions: Recent research mentions something incredible: Herbivore-Induced Plant Volatiles (HIPVs). When plants are eaten, they release chemical signals (smells) into the air. These aren’t just screams of pain; they are dinner bells. They attract predators—parasitic wasps, predatory mites, and ladybugs—that eat the pests. The research shows that Si-treated plants release more and different volatiles when attacked. They scream louder. They are better at calling for help. If you use beneficial insects in your indoor garden (like releasing Amblyseius swirskii mites to fight thrips), Silicon might actually help your good bugs find the bad bugs faster. It turns your garden into a coordinated defensive ecosystem.
3. Fungal Defense: The Anti-Mold Shield
Powdery Mildew, Botrytis, and Root Rot are the bane of high-humidity vivariums and terrariums. You create a wet environment for your tropicals, but you also create a paradise for mold.
Si creates a physical barrier to fungal hyphae penetration. When a fungal spore lands on a leaf, it tries to grow a tube (hypha) that drills into the plant cell to steal nutrients. If there is a solid layer of silica under the cuticle, the fungus hits a wall. It physically cannot drill through.
Research Proof: A study on strawberries showed that Si application reduced Powdery Mildew severity significantly.
https://www.mdpi.com/1422-0067/23/7/3803
It didn’t just stop the fungus from entering; it actually inhibited the growth of the hyphae on the surface of the leaf. Furthermore, Si treatment stimulates the production of Phytoalexins—natural antifungal compounds produced by the plant. It turns the plant cell into a toxic environment for the mold.
Part 4: The Aroid Connection – Why Your Monstera Needs This
Let’s apply this specifically to the plants we actually grow. The houseplant hobby is currently dominated by the Araceae family (Monstera, Philodendron, Alocasia, Anthurium) and epiphytes.
The Hemiepiphyte Paradox
Many of our favorite Aroids are hemiepiphytes. They start life in the ground, rooting into the soil, and then climb up trees. In the ground, they have access to mineral soil. They can uptake silica from the clay and sand. They build a strong, stiff stem to support their climb. When we bring them indoors, we cut them off from the ground. We put them in pots filled with bark and moss. We give them a moss pole, sure, but we deprive them of the mineral connection they would have had in their juvenile phase.
The “Floppy Leaf” Syndrome
Growers often complain that their Philodendron leaves are floppy, or they “melt” easily during shipping or heat waves. This is a classic symptom of weak cell walls. The leaf lacks structural rigidity. Supplementing with Si typically results in thicker, leatherier leaves within 2-3 months. The petioles (leaf stems) get stiffer. A Monstera treated with Si can hold its massive fenestrated leaves up to the light without them drooping or snapping as easily. It reduces the need for aggressive staking.
Vivarium & Paludarium Relevance
In a vivarium, you are playing a dangerous game with humidity. You want it high for the plants, but high humidity suppresses transpiration. Remember, Si uptake is driven by transpiration. If the plant isn’t drinking much water because the air is wet, it isn’t taking up much nutrient. This leads to “soft growth”—lush, green, but incredibly weak tissue that rots the moment a water droplet sits on it too long. Using a Silicon supplement in a vivarium helps counteract this. It forces the plant to build stronger cell walls even in the absence of high transpiration rates. It is the best insurance policy against the dreaded “terrarium melt” where a plant turns to mush overnight.
Part 5: Reintroducing Plant Nutrients in Soil – Don’t Buy the Wrong Stuff

You are convinced. You want to add Silicon. You go to the garden center and see a bag of “Silica Sand.” You buy it, mix it in, and… nothing happens.
Chemistry is cruel. Not all Silicon is created equal. Most silicon on Earth is Silicon Dioxide (SiO2). This is quartz. It is sand. It is incredibly stable and totally insoluble. It will take 100 years of weathering for that sand to release enough silicic acid for your plant to notice. You need Plant Available Silicon (PAS).
Here is the breakdown of the sources available to the hobbyist, and which ones actually work.
1. Potassium Silicate (K2SiO3) – The “Pro” Choice
This is the standard liquid supplement you find in hydroponic stores (brands like Pro-TeKt, Armor Si, Silica Blast). It is made by fusing silica sand with potassium carbonate at high temperatures.
- The Good: It is highly soluble. It is instantly available to the plant. It contains Potassium (K), which is a macronutrient.
- The Bad: It has a pH of about 11.3.
It is extremely alkaline. If you pour this directly into your fertilizer water, it will cause the pH to skyrocket. If the pH goes too high, other nutrients (like Calcium, Iron, and Magnesium) will “lock out” or precipitate. They turn into solids and sink to the bottom of the watering can.
- The Verdict: This is the best source for immediate results, but you must know how to mix it (we will cover this in Part 6).
2. Diatomaceous Earth (DE) – The “Natural” Slow Release
DE is the fossilized remains of diatoms, ancient algae that built shells out of glass. It is mostly amorphous silica.
- The Good: It is organic. It is cheap. It has a neutral pH. It also acts as a physical pesticide if used dry.
- The Bad: It has low solubility. It releases Si very slowly over months or years. It is not a quick fix for a deficiency.
- The Verdict: DE is an excellent soil amendment. Mixing granular DE into your potting mix provides a baseline, slow-release supply of Si that mimics the mineral soil in nature. It essentially “re-mineralizes” your sterile peat mix.
3. Rice Hulls – The Sustainable Aerator
Rice hulls are the protective husks of rice grains. Rice is a massive silica accumulator, so the husks are almost pure biogenic opal.
- The Good: They are a sustainable alternative to perlite. They add aeration. As they decompose, they release plant-available silica.
- The Bad: They decompose faster than perlite (usually within a year or two), meaning your soil will lose volume over time. They can also introduce weed seeds if not steam-sterilized.
- The Verdict: Excellent for “Living Soil” vivarium setups where decomposition is part of the cycle.
4. Stabilized Orthosilicic Acid (OSA) – The “Premium” Choice
Newer products on the market (often very expensive, sold in tiny bottles) claim to be “stabilized” Monosilicic Acid.
- The Good: They are bioavailable immediately. They are often pH neutral or acidic, meaning they don’t mess up your nutrient mix as badly as Potassium Silicate.
- The Bad: The price. You are paying a massive premium for the stabilization chemistry.
- The Verdict: While effective, they aren’t strictly necessary. Standard Potassium Silicate is just as effective if you are willing to spend 30 seconds adjusting the pH.
Part 6: Application Protocols – How to Not Kill Your Plants

Chemistry is unforgiving. If you use Silicon wrong, you can hurt your plants. Here is the field manual for safe application.
The “Golden Rule” of Mixing: Si First!
Potassium Silicate is a bully in the solution. It reacts with Calcium and Magnesium (which are in your base fertilizer or Cal-Mag supplement) to form Calcium Silicate. Calcium Silicate is… cement. It is insoluble. If you mix the concentrates together, your water will turn cloudy/milky. That cloudiness is your nutrients falling out of solution. Your plant can’t eat them anymore.
The Correct Protocol:
- Water First: Fill your watering can with plain water.
- Silicon Second: Add the Potassium Silicate. Stir well.
- pH Check: The pH will spike to 9 or 10. This is too high for plants. Use a “pH Down” product (Phosphoric Acid or Citric Acid) to bring it down to roughly 6.0–6.5.
- Fertilizer Last: Only now, once the pH is safe and the silicate is diluted, do you add your NPK or Cal-Mag.
- Result: A clear solution where everything plays nice.
Root Drench vs. Foliar Spray: The Debate
Should you water it in, or spray it on the leaves? The research gives us a clear answer.
https://www.cabidigitallibrary.org/doi/pdf/10.5555/20153013110
Root Drench:
- Pros: This is the only way to get Si into the xylem and distributed to new growth. It strengthens the stems, the roots, and the internal structure. It provides the drought and salt tolerance benefits.
- Cons: Slower to see effects on pest resistance.
- Verdict: This should be your baseline. Do this every watering or every other watering.
Foliar Spray:
- Pros: Creates an immediate barrier on the existing leaves. Excellent for stopping an active Powdery Mildew outbreak or deterring a new wave of thrips.
- Cons: Si is immobile. It does not translocate downwards. If you spray a leaf, the Si stays on that leaf. It won’t move to the roots. It won’t move to the new leaf that unfurls next week. Also, Potassium Silicate sprays can leave a white, chalky residue that looks ugly on shiny Anthurium leaves.
- Verdict: Use this as a spot-treatment for pests or disease, not as your primary nutritional method.
Dosage
- Maintenance: 0.5 mL per Liter of water (check your specific bottle, aiming for about 50ppm Si).
- Stress/Pest Outbreak: Increase to 1.0–1.5 mL per Liter.
- Warning: Potassium Silicate adds Potassium (K). If you use a lot of it, you might need to back off your main fertilizer slightly to avoid Potassium toxicity, although this is rare.
Part 7: Deep Dive – How Microbes Cycle Plant Nutrients in Soil
This is the cutting edge of indoor gardening. We used to think of nutrients as simple chemicals. Now we know it’s a biological dance involving billions of bacteria and fungi. Studies highlight the importance of the microbial community in substrates.
https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2024.1347745/full
Does Silicon hurt Microbes?
No. In fact, it might help.
- pH Buffering: By stabilizing soil pH (if used correctly), it creates a better environment for bacteria.
- Synergy: In nature, there are “Silicate Solubilizing Bacteria” that eat rocks and release silicon. In a potting mix with Diatomaceous Earth, adding beneficial microbes (like Bacillus species found in products like Hydroguard or Great White) can help break down the DE and release the Si faster for the plant.
- Biochar Interaction: If you use Biochar (another hot topic in sustainable gardening), adding Silicon is synergistic. Biochar acts as a coral reef for microbes—it gives them a home. Silicon reinforces the plant structure. Together, they create a “super soil” that mimics the incredibly fertile Terra Preta soils of the Amazon.
The “Living Soil” Vivarium: If you have a bioactive vivarium with isopods (roly-polys) and springtails, you need to be careful with dry Diatomaceous Earth. Dry DE is a mechanical insecticide; it shreds the exoskeleton of insects. It will kill your clean-up crew if you dust it everywhere. However, wet DE mixed into the soil is generally safe. Once it is wet, it isn’t dusty/abrasive in the same way. It becomes part of the soil matrix. Potassium Silicate applied via root drench is 100% safe for bioactive crews, as it absorbs into the plant and doesn’t sit on the soil surface as a killing powder.
Part 8: The Practical “Recipe” for the Ultimate Indoor Mix

Based on the research reviewed (peat alternatives + biochar + silicon + microbes), here is the “Scientifically Optimized” substrate for Aroids and Tropicals. This mix addresses drainage, nutrient retention, and the critical silicon deficiency.
The Base (Structure & Air):
- 40% Coco Chips (or Pine Bark): Provides the chunky drainage that Aroid roots need to breathe.
- 20% Coco Coir (or Peat): Provides water retention. (Coir is preferred as it is more sustainable, though it needs Calcium buffering).
- 20% Perlite or Pumice: Inert aeration to prevent compaction.
The “Superchargers” (The Science Part):
- 10% Biochar:
https://www.frontiersin.org/journals/horticulture/articles/10.3389/fhort.2025.1568055/full
Biochar increases Cation Exchange Capacity (CEC). It acts like a battery, holding onto fertilizer nutrients so they don’t wash away with the first watering. It also houses your beneficial microbes.
- 5% Worm Castings: Provides enzymes, immediate biology, and gentle organic nutrients.
- 5% Diatomaceous Earth (Granular/Aggregate): This is your Slow-Release Silicon. Try to find the “aggregate” size (often sold as oil absorbent or aquatic soil) rather than the fine flour. It holds water and slowly dissolves over years, constantly re-mineralizing the mix.
https://blog.epminerals.com/diatomaceous-earth-indoor-gardening-and-hydroponics
The Watering Routine:
- Water with a weak nutrient solution that includes Potassium Silicate (pH adjusted) every watering.
- This ensures a constant supply of Monosilicic Acid for the Lsi transporters to grab.
- Think of it as “Vitamin Water” for your plants.
Part 9: Troubleshooting & Myths
Myth: “Silica makes plants brittle.”
Fact: False. It makes them stiff, not brittle. “Brittle” implies they snap easily. Si-fortified tissues are actually tougher and harder to snap. However, they are more rigid. If you are trying to train a climbing plant around a trellis, the stems will be less flexible. Pro Tip: If you plan to bend a plant significantly, stop Si supplementation for a week or two beforehand to let the new growth come in softer.
Myth: “I can just put some quartz crystals in the pot for energy.”
Fact: This is “crystal healing” for plants. It does nothing chemically. Quartz is far too stable to release silicon in a human lifetime. You need soluble silicate. Save the crystals for your bookshelf.
Problem: “My nutrient solution turned into milk.”
Cause: You broke the Golden Rule. You mixed Silicate concentrate with Cal-Mag or fertilizer concentrate. Fix: Dump it. You precipitated the nutrients. They are now effectively sand. Start over, dilute the silicate first, lower the pH, then add the rest.
Problem: “My leaves have white spots after spraying.”
Cause: Residue. Potassium Silicate dries into a thin layer of glass/salt. It’s harmless armor, but it looks like hard water stains. Fix: It’s a cosmetic issue. You can wipe it off with a damp cloth, but you are wiping off the protection. Use a lower concentration for foliar sprays (0.25 mL/L) to minimize visible residue, or stick to root drenching if you are picky about aesthetics.
Part 10: Conclusion – The Future is Fortified
The era of “NPK-only” gardening is over. It’s a relic of the 20th century. The research from the last five years has undeniably proven that Silicon is not a luxury; it is a fundamental component of plant health that we have accidentally deleted from our indoor gardens.
We took plants out of the Earth (rich in silica) and put them in pots of coconut husk (silica-free). We made them weak. Then we wondered why they died when we went on vacation or why thrips destroyed them so easily.
By reintroducing Silicon—whether through Potassium Silicate drenches or Diatomaceous Earth amendments—we are restoring the “Glass Armor” that these plants evolved to possess. We are creating plants that are structurally sound, drought-resilient, and pest-resistant.
You don’t need a green thumb. You need biochemistry. And the biochemistry says: Add Silicon.
Key Takeaway for the Everyday Gardener
Go buy a bottle of “Silica” or “Potassium Silicate” from the hydroponics store. (Don’t worry about the brand; look for the active ingredient). Add a tiny amount (0.5 mL/L) to your watering can every time you water. Adjust the pH if you can.
Do this for 3 months. Take a “Before” photo of your Monstera. Take an “After” photo. Feel the leaves. You will never go back to “naked” water again.
Summary Data Tables
Table 1: Benefits of Silicon by Stress Type
| Stress Category | Specific Stressor | Silicon Mechanism | Observed Benefit |
|---|---|---|---|
| Abiotic | Drought | Cuticle-Silica Double Layer | Reduced transpiration; delayed wilting |
| Abiotic | Salt / Fertilizer Burn | Ion Compartmentalization | Exclusion of Na+ ions; reduced leaf burn |
| Abiotic | Heavy Metals | Cell Wall Binding | Immobilization of Cd, Pb, Mn |
| Biotic | Chewing Pests | Phytolith Formation | Mandibular wear; reduced feeding |
| Biotic | Sucking Pests (Thrips) | Cell Wall Hardening | Resistance to stylet penetration |
| Biotic | Fungi (Mildew) | Physical Barrier | Inhibition of hyphal penetration |
Table 2: Comparison of Silicon Sources
| Source | Composition | Solubility | pH Impact | Best Application |
|---|---|---|---|---|
| Potassium Silicate | K2SiO3 | High (Immediate) | Very High (Alkaline) | Hydroponics, Root Drench |
| Diatomaceous Earth | Amorphous Silica | Low (Slow Release) | Neutral | Soil Amendment (Mix in) |
| Rice Hulls | Biogenic Silica | Medium (Decay) | Neutral | Aeration / Soil Mix |
| Stabilized OSA | Orthosilicic Acid | High (Immediate) | Acidic/Neutral | Foliar Spray |
Table 3: The “Supercharged” Soil Mix Ratio
| Component | Role | Recommended % | Notes |
|---|---|---|---|
| Coco Chips / Bark | Drainage | 40% | Base structure |
| Coco Coir / Peat | Retention | 20% | Water holding |
| Perlite / Pumice | Aeration | 20% | Inert aeration |
| Biochar | Nutrient Battery | 10% | Increases CEC; microbe habitat |
| Diatomaceous Earth | Si Source | 5% | Use aggregates; slow Si release |
| Worm Castings | Biology | 5% | Enzymes and biology |

Leave a Reply