Summary
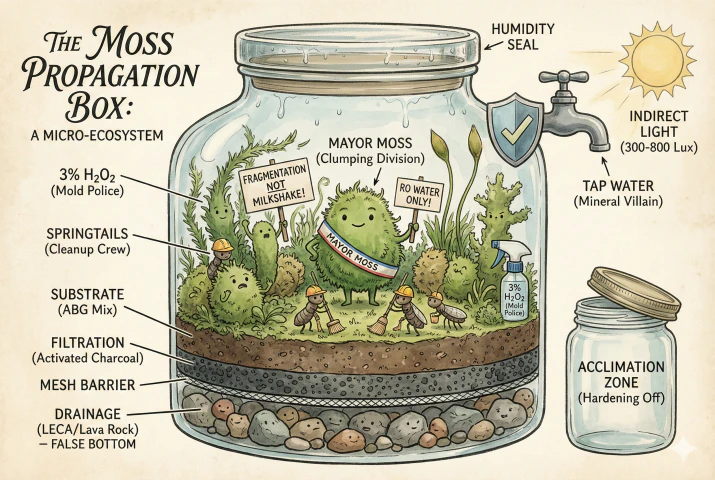
- The moss propagation box is a sealed micro-ecosystem that replicates the high-humidity “boundary layer” of a forest floor using a deep, transparent vessel to stabilize temperature and moisture.
- Success relies on a precise vertical structure involving a drainage layer (LECA/Lava Rock), a mesh barrier to prevent clogging, activated charcoal for filtration, and a nutrient-poor substrate like the ABG mix.
- Long-term health is maintained by strictly using RO or rainwater to prevent mineral toxicity, introducing Springtails to consume mold, and providing indirect light (300–800 Lux).
Key Takeaways
- Structural Integrity: The system must utilize a “false bottom” (drainage layer) separated from the soil by a fiberglass mesh. This prevents the substrate from becoming an anaerobic bog, which is fatal to moss rhizoids.
- Hydrology & Chemistry: Mosses absorb water directly through their leaves, making them hypersensitive to chemicals. You must use Reverse Osmosis (RO) water or rainwater; tap water contains chlorine and minerals that cause “mineral burn” and kill beneficial bacteria.
- Propagation Technique: Avoid the viral “moss milkshake” method (blending moss with dairy), as it causes bacterial spoilage. Instead, use fragmentation (chopping moss) for spreading or division (plug planting) for clumping species.
- Bioactive Maintenance: Introduce Springtails (Collembola), tiny arthropods that act as a natural cleanup crew by eating mold and decaying organic matter. If mold persists, spot-treat with a 3% Hydrogen Peroxide solution.
- Acclimation: Moss grown in 100% humidity cannot be instantly moved to a dry room. It requires a “hardening off” period where the box lid is gradually opened over 1–2 weeks to allow the plant cuticle to thicken.
1. Introduction: The Micro-Ecosystem Approach to Bryophyte Propagation
The cultivation of bryophytes—specifically mosses—within a controlled environment represents a synthesis of horticultural science, environmental physics, and ecosystem management. Unlike vascular plants, which rely on extensive root systems for nutrient and water uptake, mosses utilize a more primitive yet highly specialized physiology that makes them uniquely suited for, yet distinctively challenging in, propagation. The “Moss Propagation Box” is not merely a container for growing plants; it is a sealed or semi-sealed atmospheric enclosure designed to replicate the high-humidity, stable-temperature conditions of the forest floor boundary layer where these cryptogams thrive.
This report provides an exhaustive analysis of the reconstruction of a moss propagation system. It expands upon foundational instructional texts by integrating advanced insights into substrate chemistry, hydrological cycles within closed systems, and the biological imperatives of Leucobryum, Hypnum, and other common terrarium mosses. The objective is to guide the practitioner—whether a novice seeking to green a tabletop or a professional creating stock for bioactive vivariums—through the creation of a self-sustaining propagation vessel.
Bryophytes are poikilohydric organisms, meaning they have little control over their internal water content and rely heavily on the external environment to maintain hydration. This fundamental biological trait dictates every design choice in the propagation box, from the depth of the container to the chemistry of the water used. By understanding the “why” behind these requirements, the cultivator moves beyond rote instruction to a nuanced understanding of plant physiology.
2. Structural Enclosure and Environmental Parameters
The selection of the vessel dictates the gas exchange rate, light penetration, and humidity retention of the system. While a simple plastic box may suffice for basic survival, optimizing growth rates and preventing pathogen proliferation requires a deeper understanding of the physics of the enclosure.
2.1 The Propagation Vessel: Material Physics and Geometry
The ideal container is a transparent box with a transparent lid to allow light to penetrate. However, the specific optical and thermal properties of the material play a significant role in the success of the propagation.
2.1.1 Transparency and Light Transmission
Photosynthetically Active Radiation (PAR) must penetrate the lid to reach the moss colonies. Clear polypropylene (storage bins) or polycarbonate containers are standard. Glass terrariums offer superior clarity and scratch resistance but are heavier and often lack the airtight seal required for high-humidity propagation unless specifically designed for it.
- Insight: One must consider the refraction and diffusion of light. A textured plastic lid may diffuse light, which is actually beneficial for mosses that naturally grow in the dappled shade of a forest canopy. Direct, focused sunlight through clear glass can act as a magnifying lens, raising internal temperatures to lethal levels (the greenhouse effect) within minutes.
2.1.2 The Thermodynamics of “Headroom”
A “deep box” is recommended not only to accommodate the drainage and substrate layers (which may total 2–4 inches) but to provide an “atmospheric buffer” above the moss.
- Thermal Mass: A larger volume of air stabilizes temperature fluctuations. If the box is exposed to a thermal spike (e.g., a ray of sun or a rise in room temperature), a larger air volume absorbs this energy with a smaller rise in overall temperature compared to a shallow box.
- Condensation Dynamics: In a sealed system, water evaporates from the substrate and condenses on the coolest surfaces—typically the lid and walls. In a shallow container, heavy condensation on the lid is physically close to the moss. Large droplets can bridge the gap or drip incessantly onto the moss foliage. While moss loves humidity, constant liquid saturation of the phyllids (leaves) blocks gas exchange (CO2 uptake), leading to tissue necrosis and rot. Adequate headroom ensures a moisture gradient: saturated air at the substrate level, but sufficient distance to prevent direct waterlogging from condensation rain.
2.2 The Closed System Water Cycle
In a sealed propagation box, water enters a cycle of evaporation, condensation, and precipitation. Water evaporates from the substrate and transpires from the moss, condenses on the cooler walls and lid, and eventually drips back down.
- Humidity Targets: Moss propagation generally requires relative humidity (RH) between 70% and 90%. This mimics the boundary layer of air immediately adjacent to the damp soil in a forest, where evaporation is suppressed, and mosses can keep their stomata-like pores open for photosynthesis without losing turgor pressure.
- Ventilation: While high humidity is essential, total stagnation can lead to fungal pathogens. Some protocols suggest drilling small holes or opening the lid periodically (e.g., once a week) to allow gas exchange (CO2 replenishment and O2 release). However, for the purpose of establishing a propagation box, a sealed environment is often maintained for weeks to induce the “greenhouse effect” that accelerates protonemal growth, provided clean materials are used.
3. The Benthic Zone: Drainage and Separation Layers
The foundational layer of any functional terrarium or propagation box is the drainage layer, often referred to as the “false bottom”. Its purpose is hydraulic separation: it creates a reservoir for excess water, ensuring that the soil layer above remains moist but not saturated.
3.1 Layer 1: The Drainage Reservoir
The original text prescribes a gravel layer of approximately 0.5 inches (1.3 cm). This depth provides a safety margin; if the user accidentally overwaters, the excess liquid pools here rather than turning the substrate into an anaerobic bog.
3.1.1 Material Selection: Gravel vs. LECA vs. Lava Rock
While gravel is functional, modern horticultural practices offer superior alternatives based on porosity and weight.
| Material | Porosity | Weight | Biological Surface Area | Verdict |
|---|---|---|---|---|
| River Gravel | Low | High | Low | Acceptable. Standard for beginners but heavy and offers little biological filtration. |
| LECA (Clay Balls) | High | Low | High | Superior. Wicks moisture to aid humidity; houses beneficial bacteria. |
| Lava Rock | High | Medium | Very High | Excellent. Natural aesthetic; creates anaerobic zones for nitrate reduction deep inside stones. |
- Anaerobic Risks: Anaerobic conditions in the substrate lead to the production of hydrogen sulfide, which is toxic to plant rhizoids and promotes root rot. By elevating the soil above the standing water table using this drainage layer, aerobic conditions are maintained in the root zone.
- Preparation: Regardless of the material, it is imperative to rinse the aggregate thoroughly. Stone dust and silt can migrate upward into the mesh or form a sludge at the bottom that clogs the drainage voids.
3.2 Layer 2: The Mesh Barrier
Separating the drainage layer from the substrate is critical to the longevity of the system. Without a barrier, fine soil particles will wash down into the gravel under the influence of gravity and watering, clogging the voids and eliminating the drainage capacity.
3.2.1 Material Science: Carbon Fiber vs. Fiberglass
The “Carbon-Fiber Mesh” mentioned in the source text is a high-grade option, but fiberglass window screening is the industry standard for this application due to its availability, durability, and rot resistance.
- Carbon Fiber: Extremely durable and chemically inert. It is often used in high-end vivariums but may be cost-prohibitive for a simple propagation box.
- Fiberglass Window Screen: The most practical choice. It is rot-proof, chemically neutral, and has a fine aperture that retains soil while allowing water to pass.
- Avoid Organic Fabrics: Materials like cotton, burlap, or cheesecloth will decompose rapidly in the warm, humid environment of a propagation box. Their breakdown can cause the substrate layer to collapse into the drainage layer, ruining the setup.
3.3 Layer 3: The Charcoal Layer
Directly above the mesh (or sometimes mixed into the drainage layer), a layer of charcoal is applied. This layer serves as a chemical filter and a biological buffer.
3.3.1 Adsorption and “Sweetening” the Soil
- Activated Carbon vs. Horticultural Charcoal:
- Activated Carbon: Processed at high temperatures to maximize surface area and adsorption (the adhesion of atoms/molecules to a surface). It is highly effective at trapping volatile organic compounds (VOCs), toxins, and odors. However, research indicates its adsorption capacity is finite, becoming inert after a few months of saturation.
- Horticultural Charcoal: Often lumpier and less processed than activated carbon. While less efficient at chemical filtration, it persists indefinitely as a soil amendment, improving drainage and porosity.
- The “Soil Sweetening” Function: In the context of a closed box, “sweetening” refers to the prevention of acidification and foul odors caused by stagnation. Charcoal absorbs the byproducts of decomposition. While some argue that in a fully bioactive system microbes handle waste effectively enough to render charcoal obsolete, in a propagation box—where water flow is minimal and the system is not fully established—charcoal acts as a critical “safety net” to absorb initial impurities and stabilize the environment.
4. The Substrate: Edaphic Factors in Moss Culture
The substrate is the biological engine of the propagation box. Unlike standard potting soil, which is often too dense and nutrient-rich (risking fertilizer burn), moss substrates must be low-nutrient, acidic to neutral (depending on species), and structurally stable to prevent compaction over time.
4.1 The ABG Mix Standard
The “ABG Mix” (developed by the Atlanta Botanical Garden) is considered the gold standard for bioactive terrariums and works exceptionally well for moss propagation. It is designed to last for years without breaking down into an anaerobic sludge. A typical recipe includes:
- 2 parts Tree Fern Fiber: Provides structural integrity and slow decay.
- 1 part Sphagnum Peat Moss: Acidifies the mix and retains water.
- 2 parts Orchid Bark: Adds bulk, aeration, and drainage.
- 1 part Sphagnum Moss: Increases hydration capacity.
- 1 part Charcoal: Sweetens the mix.
Modifications for Propagation Boxes: For pure moss propagation, a simpler mix is often sufficient if ABG ingredients are unavailable. Many growers use a base of:
- Sphagnum Peat Moss or Coco Coir: Peat is preferred for acid-loving mosses like Leucobryum glaucum (Cushion Moss). Coco coir is more neutral and sustainable but may require rinsing to remove salts.
- Perlite/Pumice: Essential for aeration. Moss rhizoids require oxygen; a compacted soil will kill the colony.
- Warning on Potting Soil: Do not use “All Purpose Potting Soil” with added fertilizers (e.g., Miracle-Gro). The high nitrogen content can chemically burn moss and promote rapid algal and mold growth that outcompetes the slow-growing bryophytes.
4.2 Chemical and Physical Soil Properties
- pH Requirements:
- Acidophiles: Leucobryum glaucum (Cushion Moss) and Polytrichum thrive in acidic substrates (pH 5.0–6.0). A peat-heavy mix is ideal here.
- Neutrophiles: Thuidium delicatulum (Fern Moss) is often found on neutral soil or logs. A mix with less peat and more bark/coir is appropriate.
- The Substrate-Rhizoid Interface: The success of moss propagation relies heavily on the contact zone between the moss and the soil. Unlike vascular plants that can push roots deep into soil in search of water, moss rhizoids are superficial. If the top layer of the substrate dries out, the moss desiccates. This is why the instruction to Pre-mist the soil is chemically significant. Dry organic matter (like peat) often develops hydrophobicity (water repellency). Pre-misting breaks the surface tension, creating a continuous water column from the substrate to the moss.
5. Biological Installation: Planting and Establishment
With the physical structure (Box → Gravel → Mesh → Charcoal → Substrate) complete, the focus shifts to the biological elements. This phase is where the art of aquascaping meets the science of regeneration.
5.1 Preparation and Sourcing
- Pre-mist: As instructed, the soil must be moistened before planting. The goal is “moist, not soggy”—when squeezed, a drop or two of water should appear, but it should not run in a stream.
- Sourcing Ethics: Never harvest moss from the wild without permission and knowledge of local conservation laws. Over-harvesting damages ecosystems significantly. Sustainable fragmentation (taking small pieces to propagate) is preferred over removing whole colonies.
5.2 Propagation Techniques: Fragmentation vs. Division vs. The “Milkshake”
Mosses exhibit totipotency, meaning nearly any cell has the potential to regenerate into a new plant. This biological trait allows for various propagation methods.
5.2.1 The Fragmentation Method (Chopping)
This is the most efficient method for spreading limited stock over a large area, particularly for sheet mosses like Hypnum.
- Tear/Cut: The moss is torn or cut into small fragments (approx. 0.5 cm).
- Spread: These fragments are sprinkled evenly over the prepared substrate.
- Press: They are pressed firmly into the soil to ensure contact.
- Result: Each fragment will revert to a protonemal stage (algal-like threads) before producing new leafy shoots. This method requires patience (months) but yields a dense, uniform carpet.
5.2.2 The Division Method (Plug Planting)
Best for acrocarpous (clumping) mosses like Leucobryum glaucum (Cushion Moss), which do not spread rapidly via runners.
- Isolate: Break a large cushion into smaller, quarter-sized plugs.
- Plant: Nestle these plugs into the substrate.
- Sectioning: Grouping similar mosses together (as advised in the original text) prevents aggressive growers like Hypnum from overrunning slower species like Leucobryum.
5.2.3 The “Moss Milkshake” Myth
A common internet trope involves blending moss with buttermilk or yogurt to create a “paintable” slurry.
- Research Verdict: Avoid this method. While conceptually sound (spreading spores/fragments), the introduction of sugars and proteins from dairy into a warm, humid propagation box inevitably leads to rapid bacterial and fungal spoilage. The mold blooms typically kill the moss fragments before they can establish. Professional growers rely on water-only fragmentation or division.
5.3 Planting Technique: The Importance of Contact
Moss does not have true roots to dig into the soil; it relies on rhizoids for anchoring. These structures require intimate contact with the substrate to develop. The instruction to “gently press (pat) it down” is vital. This eliminates air pockets under the moss which would otherwise act as insulation, preventing capillary action from wicking water up from the damp soil to the moss tissues.
6. Hydrology and Water Chemistry
Water quality is perhaps the single most significant variable in moss culture. Bryophytes absorb water and nutrients directly through their leaves, making them hypersensitive to dissolved solids and chemicals.
6.1 The Danger of Tap Water
- Chlorine and Chloramine: Municipal water supplies are treated with these chemicals to kill bacteria. In a propagation box, they are biocidal to the delicate cell structures of moss and the beneficial microbiome (like Springtails) that keeps the system healthy.
- Dissolved Minerals (Hard Water): Tap water often contains high levels of calcium carbonate and magnesium. Because moss leaves lack a cuticle (waxy protective layer) comparable to vascular plants, these minerals can accumulate on the leaf surface as water evaporates. This “scale” blocks stomata and inhibits photosynthesis, causing the moss to turn brown and brittle (mineral burn).
6.2 The Solution: RO and Rainwater
The recommendation to use Reverse Osmosis (RO) water or rainwater is non-negotiable for long-term success.
- Total Dissolved Solids (TDS): These sources have near-zero TDS. This prevents the “salinization” of the substrate. In a closed system, minerals never leave; they only accumulate. Using low-TDS water ensures that the osmotic balance remains favorable for the moss cells.
- Rainwater Caution: In urban areas, rainwater can be acidic or polluted. Collect from a clean catchment area after the rain has been falling for some time (washing the air of particulates).
7. The Bioactive Cleanup Crew: Springtails
A closed propagation box is a humid environment rich in organic matter—a perfect breeding ground for mold. To combat this without chemical fungicides (which would harm the moss), we introduce a biological control agent.
7.1 Enter the Springtail (Collembola)
Springtails are minute, hexapod non-insect arthropods. They are detritivores that specialize in consuming fungal hyphae (mold roots), decaying plant matter, and bacteria.
- Mechanism: They roam the substrate and moss fronds, eating mold spores before they can bloom into visible outbreaks. They effectively “clean” the moss.
- Nutrient Cycling: By consuming waste and excreting it, they mineralize nutrients, making them available to the moss in trace amounts, completing the nitrogen cycle within the box.
7.2 Culturing and Introducing Springtails
- Sourcing: Cultures are available from terrarium supply stores. They are typically sold on a substrate of charcoal or calcium-bearing clay.
- Charcoal Culture: Easy to pour water into the culture, float the springtails, and dump them into the new box.
- Clay Culture: Considered superior for breeding density. To harvest, one adds water to the clay culture, tilts it, and pours the floating insects into the propagation box.
- Inoculation: Simply pour a portion of the culture (bugs and substrate, or just the bugs via water floatation) into the propagation box. They will quickly colonize the charcoal and drainage layers.
- Maintenance: They require moisture (which the box provides) and food (mold/detritus). In a sterile setup, one might add a grain of uncooked rice or yeast occasionally to feed them until the ecosystem matures, though usually, the organic substrate provides sufficient food.
8. Maintenance and Environmental Control
While described as “low maintenance”, a propagation box requires monitoring, particularly in the early stages, to ensure the physical parameters remain within the biological tolerances of the moss.
8.1 Lighting: The “Goldilocks” Zone
Moss is often misunderstood as a “low light” plant. While it tolerates low light, it does not grow fast in it. To propagate, light must be significantly above the “compensation point” (where energy produced = energy consumed).
- Intensity: Target a light level of 300 to 800 Lux (approx. 30–80 foot-candles) for maintenance, and slightly higher for active propagation.
- Source: Indirect sunlight (e.g., a North-facing window) is ideal. Direct sun is dangerous; it acts as a magnifying glass through the clear lid, cooking the moss via the greenhouse effect.
- Artificial Light: Full-spectrum LED grow lights (6500K color temperature) are excellent. They provide the correct spectrum for photosynthesis without the radiant heat of incandescent bulbs, preventing desiccation.
8.2 Mold Management and Remediation
Even with springtails, mold can occur, especially on wooden elements or decaying moss parts.
- Spot Treatment: If mold appears, dip a Q-tip in 3% Hydrogen Peroxide and dab the mold directly. The peroxide oxidizes the fungus, killing it, and breaks down into water and oxygen, leaving no toxic residue.
- Ventilation: If mold persists, the humidity is likely too high. Open the lid for a few hours to lower the relative humidity and disrupt the fungal growth cycle.
8.3 Fertilization: The Foliar Approach
Moss requires very little nutrition. Over-fertilization is lethal.
- Foliar Feeding: Since mosses absorb nutrients through their leaves, traditional soil fertilization is inefficient and dangerous (salt buildup). If the moss looks pale after months of growth, a very dilute spray of urea-free orchid fertilizer (diluted to 1/4 or 1/8 strength) can be applied directly to the foliage.
- Frequency: Rarely. Once a month or less is sufficient. In a substrate-based propagation box, the slow breakdown of the organic soil usually provides adequate nutrition without supplementation.
9. Acclimation and Harvest
The final step in the propagation lifecycle is using the moss. Moss grown in a 100% humidity box is physiologically adapted to that environment—it has a very thin cuticle and fully open pores.
9.1 The Hardening Off Process
Moving moss instantly from the propagation box to a living room environment (40% RH) will cause “shock” and rapid desiccation.
- Gradual Ventilation: Over a period of 1–2 weeks, crack the lid of the box slightly.
- Adaptation: This lowers the humidity gradually, forcing the moss to thicken its cuticle and adjust its cellular turgor pressure.
- Transplant: Once acclimated, the moss can be moved to terrariums, vivariums, or even outdoor gardens (if species appropriate).
Conclusion
The construction of a moss propagation box is an exercise in biomimicry. By layering gravel, mesh, charcoal, and specialized substrate, the grower recreates the geology and hydrology of a forest floor. By introducing springtails, the biological balance of a decomposer ecosystem is established. And by utilizing RO water and appropriate lighting, the chemical and energetic needs of the bryophytes are met. This system, once balanced, requires minimal intervention, serving as a self-sustaining nursery for some of nature’s most ancient and resilient plants.
Summary of System Components
| Component | Recommended Material | Function |
|---|---|---|
| Container | Clear Polycarbonate/Glass | Enclosure, Light transmission, Humidity retention |
| Drainage | LECA / Lava Rock | Water reservoir, prevents anaerobic root rot |
| Barrier | Fiberglass Mesh | Separates soil from drainage, prevents clogging |
| Filtration | Horticultural Charcoal | Chemical absorption, soil sweetening |
| Substrate | ABG Mix or Peat/Bark/Pumice | Acidic growing medium, aeration, moisture retention |
| Water | RO / Rainwater | Hydration without mineral toxicity (TDS control) |
| Clean-up Crew | Springtails (Collembola) | Mold control, nutrient cycling |
| Lighting | LED (6500K) or Indirect Sun | Energy for photosynthesis (300-800 Lux) |
References & Further Watching
Visual Guide to ABG Substrate Mix:
This video provides a quick visual breakdown of the “ABG Mix” (Atlanta Botanical Garden) substrate mentioned in Section 4.1. It is useful for visualizing the consistency and ratio of tree fern fiber, peat moss, and charcoal required for a high-quality vivarium substrate.
Moss Propagation Box Walkthrough:
This guide demonstrates the full assembly of a propagation box, including the harvesting of moss and the “fragmentation” technique discussed in Section 5.2.1. It is an excellent visual companion to the step-by-step instructions in this report.
General Moss Propagation Techniques:
For those interested in the broader context of moss propagation, this video covers various methods including the “division” technique for clumping mosses and managing humidity levels during the acclimation phase discussed in Section 9.1.

Leave a Reply